 Problem 4: In the above two examples which rock is more dense? which is less dense? Problem 3: In the above two examples which rock is heavier? Which is lighter? Problem 2: You have a different rock with a volume of 30cm3 and a mass of 60g. Problem 1: You have a rock with a volume of 15cm3 and a mass of 45 g. Using the equation and the information we discussed above, try to answer some of these practice problems from Carleton College’s website. The data below is from Harper College’s website. You would then have to make sure that your sample is always equilibrated to the calibrated temperature of the pycnometer. 
With that, measurement is only valid at that certain temperature. Keep in mind that most pycnometers are calibrated for use in certain temperatures. The density is calculated based on this formula Density = (M2 − M1)/Flask Volume. API (American Petroleum Institute Index)Īnother tool that would help you measure the density of a liquid is pycnometers.Ī pycnometer is usually made up of glass and a ground stopper.Keep in mind that aside from its usual specific gravity values, a hydrometer can also be calibrated to 
It affects the density of the water because mass also increases every time you put salt in it.īy simply referring to the formula we cited before, we can already infer that salinity does affect water salinity. So when the water froze and turns into ice, it becomes denser as compared to when it was in its liquid state. In the picture above, ice floats in water.Īs we glossed over a few times, water temperature alters the water density. The water density table below is from metrologia Temp./ CĮven though ice is the solid-state of water, it is less dense than liquid water. If you look at the table below, you would see that the weight and density of the water slightly change as temperature changes. So when you boil water, it would slightly get lighter. Read more about how to correctly acknowledge RSC content.Oil is less dense than water (below 1) so it is no wonder why it floats on water.Īs we have pointed out before, the temperature can alter the density of water. Please go to the Copyright Clearance Center request page. In a third-party publication (excluding your thesis/dissertation for which permission is not required) If you want to reproduce the whole article If you are the author of this article, you do not need to request permission to reproduce figuresĪnd diagrams provided correct acknowledgement is given. Provided correct acknowledgement is given. If you are an author contributing to an RSC publication, you do not need to request permission To request permission to reproduce material from this article, please go to the Indanone-based conjugated polymers enabling ultrafast electron transfer for visible light-driven hydrogen evolution from water This study provides valuable insights into the potential of IC-based conjugated polymers for photocatalytic hydrogen evolution. 
As a result, ICTDB, photocatalysts with IC-containing structures achieved a hydrogen evolution rate of 30.0 mmol g −1 h −1, which was 11.5 times higher than that of ICFTDB, the polymer with no malononitrile substitution. Through transient absorption spectroscopy, we demonstrated that ICTDB exhibited enhanced capabilities for ultrafast electron transfer and reduced recombination effects. We investigated the correlation between the optical, electrochemical, and hydrogen evolution performances of these polymers. 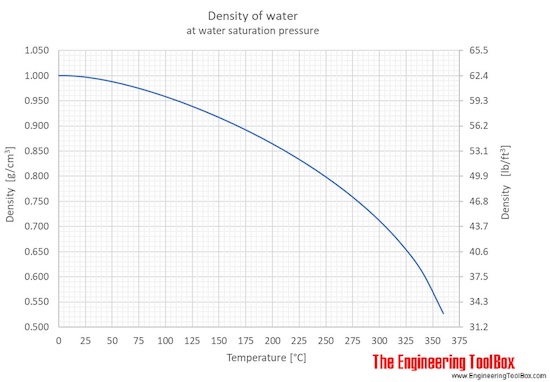
These monomers were used to synthesize polymers with varying degrees of malononitrile substitution, referred to as ICFTDB, ICTDB, and IDMTDB. In this study, we designed a series of novel IC-based monomers incorporating a dibenzothiophene- S, S-dioxide unit through Suzuki coupling. However, research on the application of IC structures in PHP is limited due to synthesis challenges. 1,1-dicyanomethylene-3-indanone (IC) has been widely used as an end group in organic photovoltaics owing to its strong electron-withdrawing ability and planarity. Photocatalytic hydrogen production (PHP) from water is a promising solution for environmental pollution due to its high energy density and the abundant availability of water and solar energy on Earth. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
